 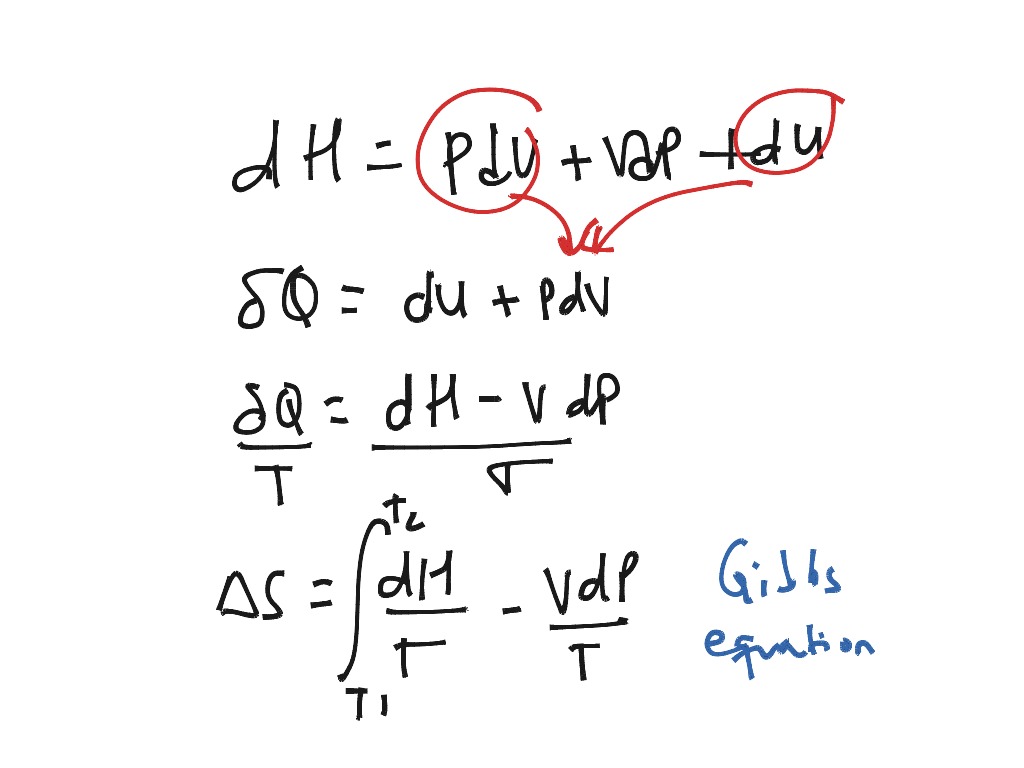
Overall, the n moles of ideal gas in the heat reservoir go from the state specified by P 1 and T P to the state specified by P. We can calculate the entropy change for the ideal-gas heat reservoir. Where q is the heat transferred in joules and ΔT is the change in temperature in Kelvin. The entire process is reversible, the entropy change for the system and the entropy change for the surroundings sum to zero: S + S 0. If there are 2 moles of reactant forming 1 mole of product, the number of states possible decreases and entropy decreases, likewise, if 1 mole of reactant forms 2 moles of product, the entropy increases.Įntropy can be calculated using the equation Example of increasing entropy: Ice melting in a warm room is a common example of increasing entropy. If a substance goes from a liquid to a solid, the molecules in the system occupy a lower number of states (think about the decrease of molecule activity in water when it freezes to ice) and therefore the entropy decreases if a substance goes from a solid to a liquid, the entropy increases. When considering whether reactions have an increase or decrease in entropy, two factors are important: The number of moles created and states of the reactants versus the products. Liquids can be considered the second most disordered, and solids can be considered the least disordered. Because molecules in gases are able to change both volume and shape, they can be considered the most disordered. We can think of the degrees of the number of different states the molecules in each can occupy. For the Gibbs thermodynamic potential G all calculations are parallel to those for F. Thermodynamics: Tds relation ideal gas, calculating entropy change for ideal gas, s-function, isentropic compression function Watch this video on YouTube The change in entropy of a system for an arbitrary, reversible transition for which the temperature is not necessarily constant is defined by modifying SQ/TSQ/T.  
Solids, liquids, and gasses all have different degrees of disorder. Show that the entropy change in the cyclic process of an ideal gas. It explains how to calculate the entropy change of heating water from 0C to 100C using two different formulas. The concept of disorder can best be described in terms of the states of matter. The various methods are divided into three main categories, the counting approach, thermodynamic integration/perturbation, and methods for calculating the. The unit of entropy is Joules per Kelvin or J/K.Įntropy is the measure of the disorder of a system and can is the energy of a system over its temperature, represented as J/K. The Second Law of Thermodynamics states that the entropy (or disorder) of a system is constantly increasing. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
